Texas Heart Institute and Rice Nanotube Fibers Move up to the Final 16

Click Here to Vote Texas Heart Institute
HOUSTON, TX – (March 16, 2020) – March Madness for the science, medicine, and technology enthusiast continues, and Texas Heart Institute and Rice advanced to the Final 16!
The discovery was published in the American Heart Association’s Circulation: Arrhythmia and Electrophysiology (Circulation A&E) as an open-access Editor’s Pick and is now completing with fifteen other biomedical advancements for the coveted Stat Madness title based on a publication of their biomedical research in the prior year.
Round 3 of the voting is another single-elimination contest, which began Monday, March 16. Round 3 voting is open until March 19 at 10:59 p.m. A popular vote decides the winner of each matchup.
The study co-led by Dr. Mehdi Razavi and Rice University chemical and biomolecular engineer Matteo Pasquali reported the creation of the promising new technology to prevent death due to cardiac arrhythmias — harnessing the conductive power of carbon nanotube fibers (CNTf).
The heart research applies a pioneering 2013 invention by Rice University chemical and biomolecular engineer Matteo Pasquali.
The conductive fibers made out of carbon nanotubes are also being studied for electrical interfaces with the brain for use in cochlear implants, as flexible antennas, and for automotive and aerospace applications. At the time of the study, co-lead author, Mark D. McCauley, MD, Ph.D. was a fellow at the Texas Heart Institute in the ACGME-accredited Texas Heart Institute Clinical Cardiac Electrophysiology Fellowship sponsored by Baylor College of Medicine and located at THI and Baylor St. Luke’s Medical Center in Houston, Texas. Dr. McCauley carried out many of the clinical experiments as a postdoctoral fellow at THI. He is now an assistant professor of clinical medicine at the University of Chicago in Illinois, specializing in heart rhythm management treating patients with atrial fibrillation, atrial flutter, heart failure, and ischemic heart disease. The co-authors were Colin Young and Julia Coco of Rice; Brian Greet of THI and Baylor St. Luke’s Medical Center; Marco Orecchioni and Lucia Delogu of the Città della Speranza Pediatric Research Institute, Padua, Italy; Abdelmotagaly Elgalad, Mathews John, Doris Taylor, and Luiz Sampaio, all of THI; and Srikanth Perike of the University of Illinois at Chicago. Pasquali is the A.J. Hartsook Professor of Chemical and Biomolecular Engineering, a professor of materials science and nanoengineering and chemistry.
Why is this innovation so promising? Patients often form scar tissue after a heart attack, and this resulting scar disrupts the normal conduction in the heart. Patients with congestive heart failure or dilated cardiomyopathy also experience conduction problems.
This disruption can lead to lethal ventricular arrhythmias that currently can only be treated by cardiac defibrillators. Cardiac defibrillators can reset the heart, but do not fix the conduction problem and are very painful. The traditional methods of pacing —like pacemaker leads— cannot bridge over scarred heart muscle to prevent disruptive arrhythmias from solving this problem.
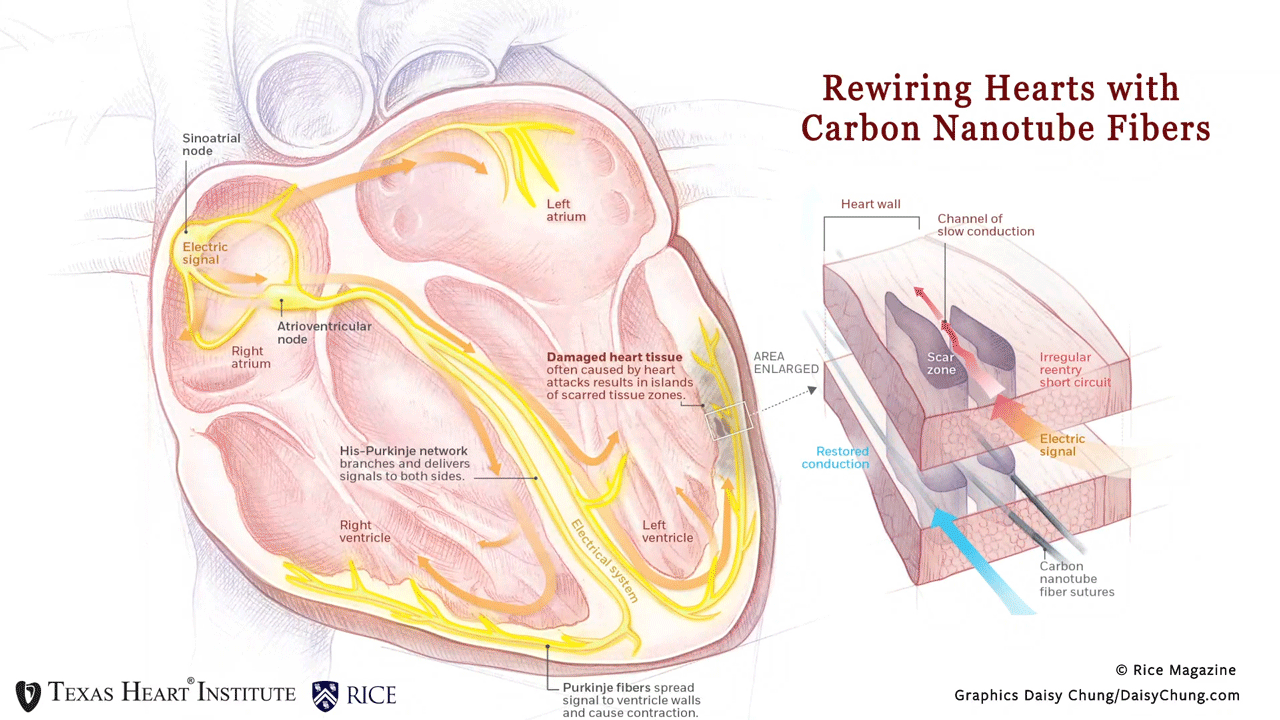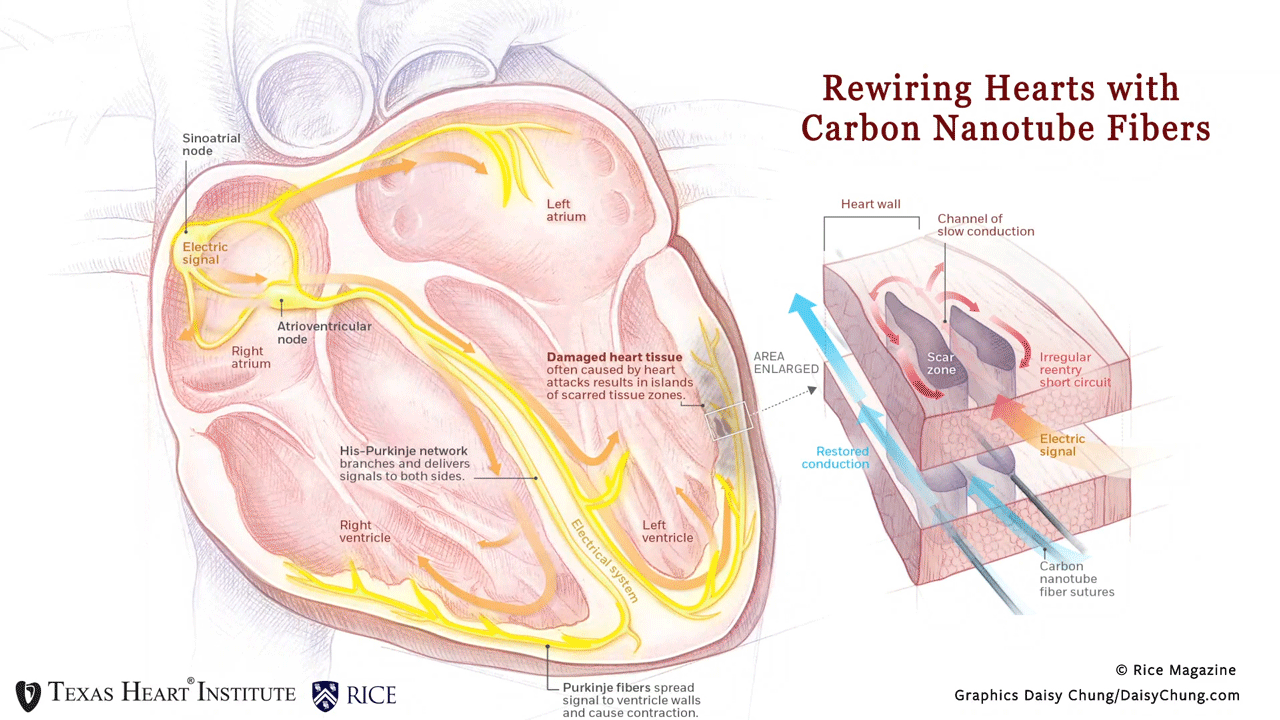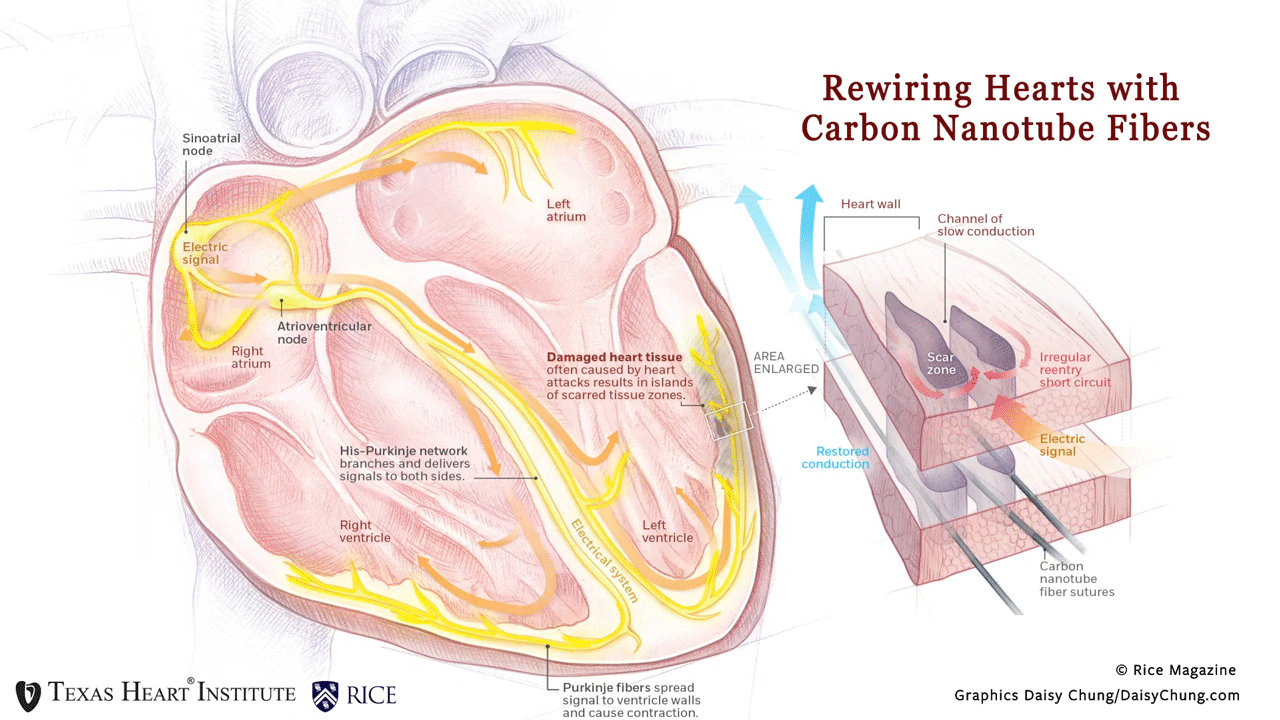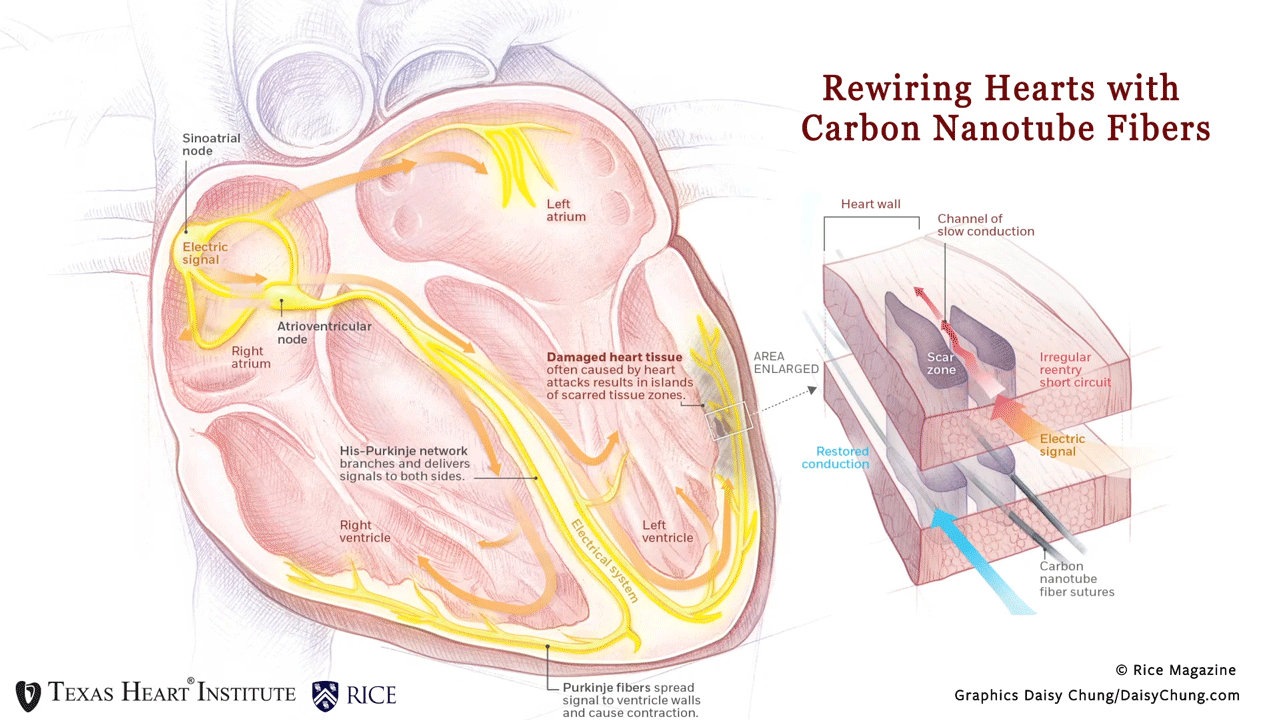
“By using thread-like carbon nanotube fibers with high flexibility and high electrical conductivity, we have created a never-before-seen paradigm of pacing over scar tissue and restored native conduction in an in vivo model with a synthetic material.,” said Dr. Mehdi Razavi. In this article, we have bridged across scar tissue and normalized conduction in both small and large preclinical research models. “Not only will this groundbreaking work create an opportunity to improve the treatment of millions of people with ventricular arrhythmias, but this therapy has the potential to improve the treatment of almost any cardiac arrhythmia,” added Dr. Razavi.
Follow the Contest on Twitter at #STATMadness. Vote for Texas Heart Institute/Rice University!

Click Here to Vote Texas Heart Institute
Rewiring Hearts With Nanotubes Soft fibers bridge gap to restore healthy beat. (n.d.). Rice Magazine Winter 2020, 25. Graphic by Daisy Chung
Read the paper: McCauley MD, Vitale F, Yan JS, et al. In vivo restoration of myocardial conduction with carbon nanotube fibers Circulation: Arrhythmia and Electrophysiology. 2019;12 https://www.ahajournals.org/doi/10.1161/CIRCEP.119.007256
This work was supported by the American Heart Association (15CSA24460004), Welch Foundation (C-1668), Air Force Office of Scientific Research (FA9550-09-1-0590 and FA9550-15-1-0370), the National Institutes of Health (K08 HL130587), and generous donation by Mr Louis Magne (Dr McCauley).


