Texas Heart Institute’s Molecular Cardiology Research Laboratory (MCRL) is a unique team of researchers and scientists dedicated to leveraging their expertise in small molecule therapeutics to develop novel techniques to diagnose, treat and prevent cardiovascular disease.
Molecular Cardiology Research
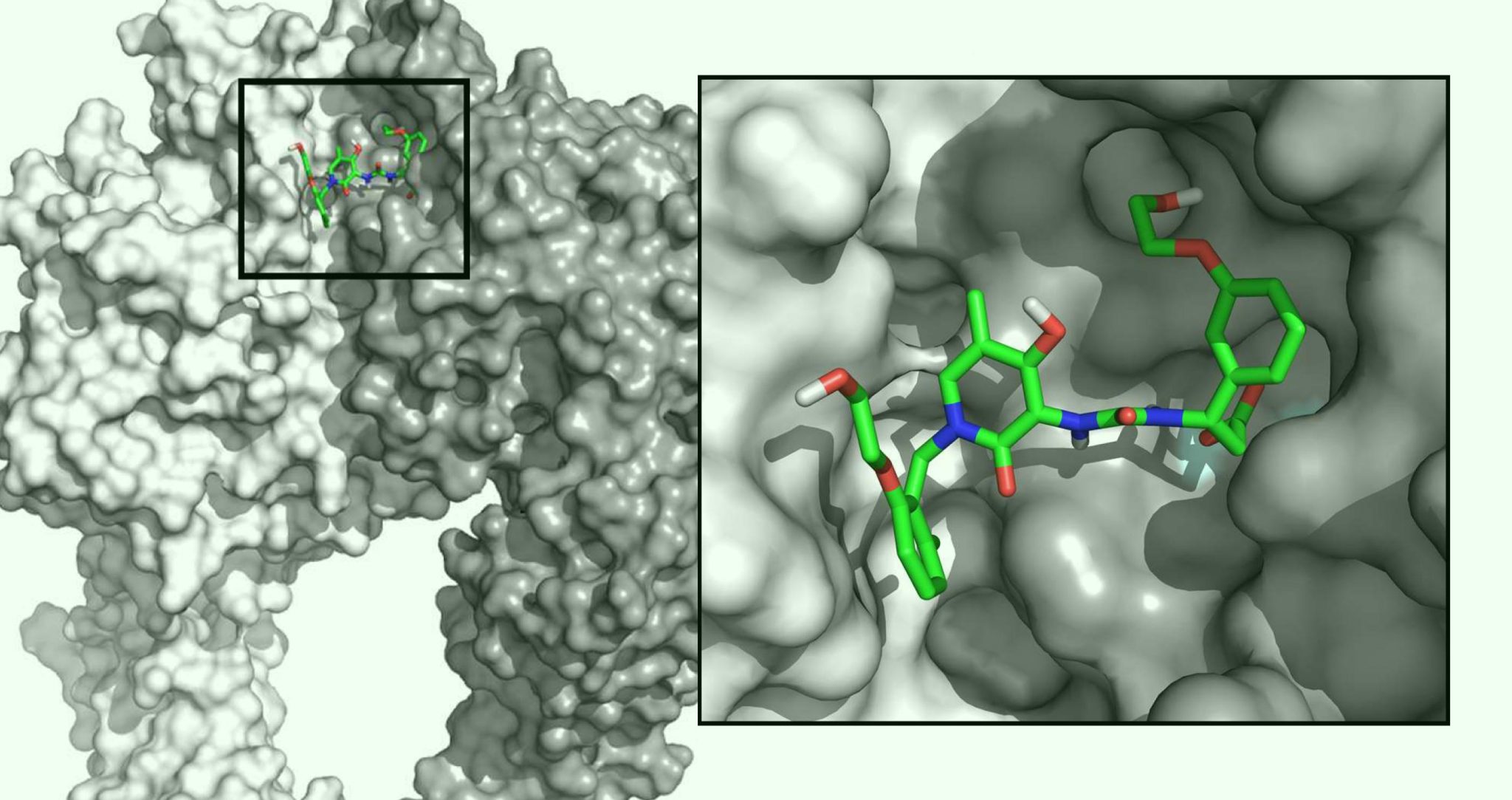
We are pushing boundaries in the research and development of small molecule and cell-based therapeutics.
The Molecular Cardiology Research Laboratories seek to better understand the molecular mechanisms of heart disease while pushing boundaries in the research and development of small molecule and cell-based therapeutics. The team leverages their expertise in molecular biology to discover ways to improve the treatment of various cardiovascular diseases.

Our Team
Our mission is to improve the treatments available for patients suffering from cardiovascular disease by combining basic research aimed at understanding the molecular mechanisms of heart disease with the discovery of innovative small molecule and cell-based therapeutics.
Current Projects
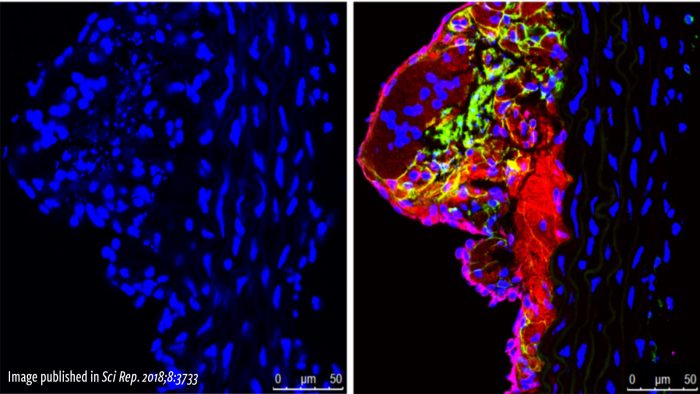
The team is leveraging their expertise in molecular biology to discover many new ways to improve the treatment of various cardiovascular diseases.
Recent News
Discoveries could lead to earlier detection of diseass to promote lifesaving interventions before a heart attack or stroke.




